Kirjoita osake tai kryptovaluutta hakupalkkiin saadaksesi yhteenvedon

Blueprint Medicines Corp
2L9Blueprint Medicines Corporation, a precision therapy company, develops medicines for genomically defined cancers and blood disorders in the United States and internationally. The company is developing AYVAKIT for the treatment of systemic mastocytosis (SM) and gastrointestinal stromal tumors; BLU-263, an orally available, potent, and KIT inhibitor for the treatment of indolent SM, and other mast cell disorders. It is also developing GAVRETO for the treatment of RET fusion-positive non-small cell lung cancer, altered thyroid carcinoma, and medullary thyroid carcinoma; BLU-945 for the treatment of epidermal growth factor receptor driven non-small-cell lung carcinoma (NSCLC); and BLU-451 to treat NSCLC in patients with epidermal growth factor receptor gene (EGFR) exon 20 insertion mutations. In addition, the company is developing BLU-782, for the treatment of fibrodysplasia ossificans progressive; BLU- 222 to treat patients with cyclin E aberrant cancers; and BLU-852 for the treatment of advanced cancers. It has collaboration and license agreements with Clementia Pharmaceuticals, Inc.; Proteovant Therapeutics; CStone Pharmaceuticals; Genentech, Inc.; Hoffmann-La Roche Inc.; and Zai Lab (Shanghai) Co., Ltd. The company was formerly known as Hoyle Pharmaceuticals, Inc. and changed its name to Blueprint Medicines Corporation in June 2011. Blueprint Medicines Corporation was incorporated in 2008 and is headquartered in Cambridge, Massachusetts. Address: 45 Sidney Street, Cambridge, MA, United States, 02139
Analytics
WallStreetin tavoitehinta
125.17 USDP/E-suhde
–Osinkotuotto
–Kuluva vuosi
Edellinen vuosi
Nykyinen vuosineljännes
Viimeinen vuosineljännes
Kuluva vuosi
Edellinen vuosi
Nykyinen vuosineljännes
Viimeinen vuosineljännes
Avainluvut 2L9
Osinkoanalytiikka 2L9
Osinkojen kasvu 5 vuoden aikana
–Jatkuva kasvu
–Maksusuhde 5 vuoden keskiarvo
–Osinkohistoria 2L9
Osakkeen arvostus 2L9
Talousasiat 2L9
| Tuloksia | 2019 | Dynamiikka |



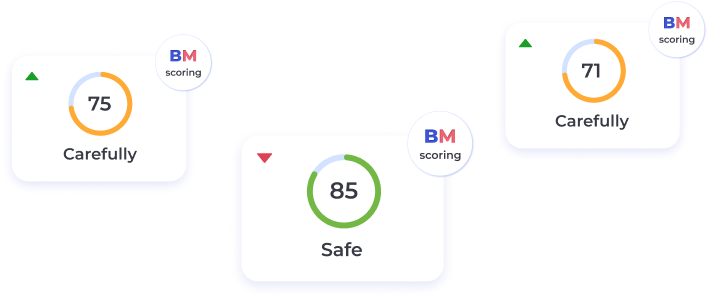


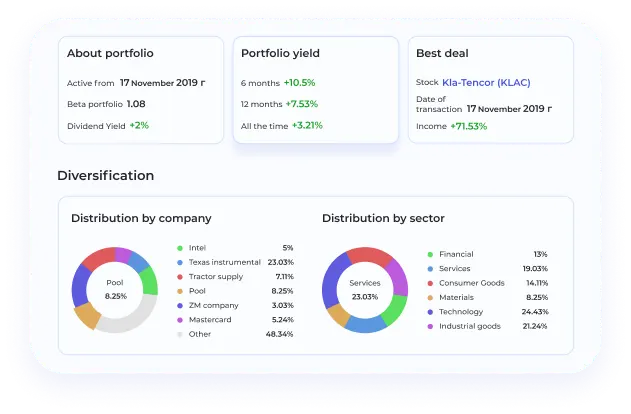


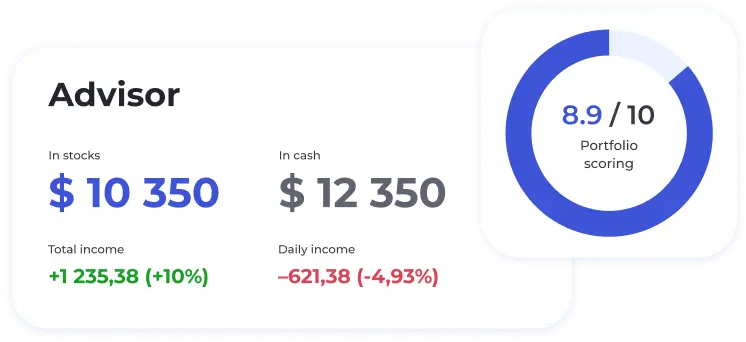
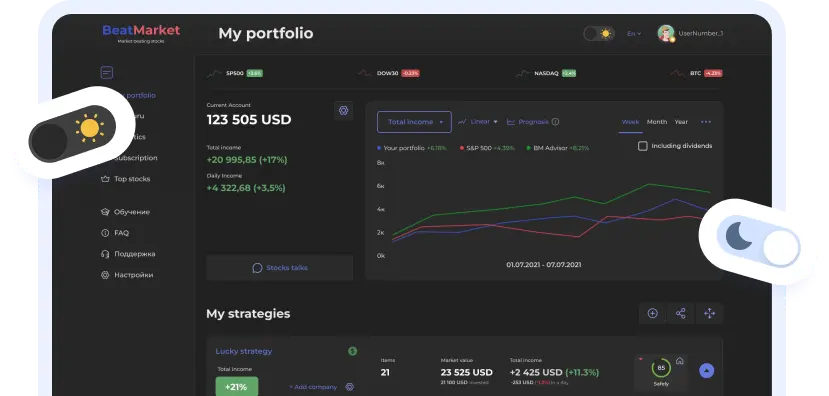



 BeatStart
BeatStart