ATAI Life Sciences BV
9VCAtai Life Sciences N.V., a clinical-stage biopharmaceutical company, develops and invests in various therapeutics to treat depression, anxiety, addiction, and other mental health disorders. Its product candidates include COMP360, a proprietary psilocybin which is in Phase 3 program; BPL-003, an intranasal formulation that is in Phase 2a and 2b clinical studies; TRD; VLS-01, an transmucosal film in Phase 1b clinical studies; and ELE-101, a serotonergic psychedelic that is in Phase 1/2a study, for the treatment of treatment resistant depression. The company also develop IBX-210, an intravenous formulation of ibogaine, and DMX-1002, an oral formulation of ibogaine indicated for the treatment of opioid use disorder; EMP-01, an oral formulation of an R-MDMA derivative being developed for the treatment of post-traumatic stress disorder; and EGX-A and EGX-B, psychedelic-like with novel, non-tryptamine structures with differentiated 5-HT receptor pharmacology. In addition, it offers RL-007, an orally bioavailable compound, which is a pro-cognitive neuromodulator for cognitive impairment associated with schizophrenia; and GRX-917, a deuterated etifoxine for the treatment of generalize anxiety disorder. Further, the company develops PCN-101, a subcutaneous formulation of R-ketamine, as a therapy for psychiatric indications initially focused on TRD; KUR-101, a formulation of deuterated mitragynine for the treatment of OUD; and RLS-01, indicated for treatment resistant depression. ATAI Life Sciences N.V. was founded in 2018 and is headquartered in Berlin, Germany with offices in New York and London. Address: Wallstrasse 16, Berlin, Germany, 10179




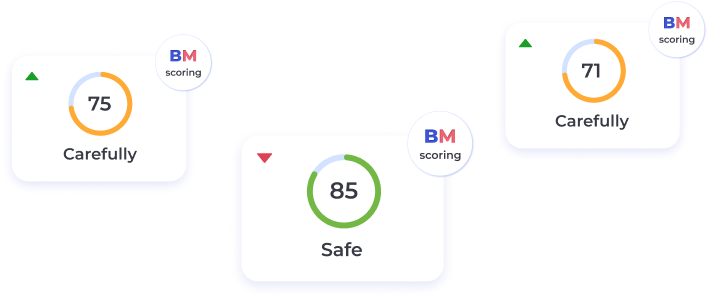


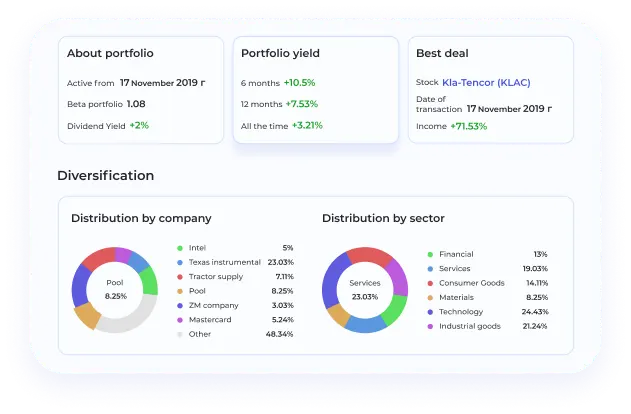


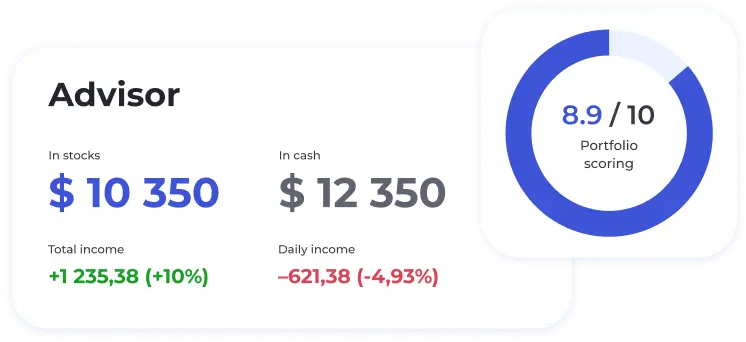
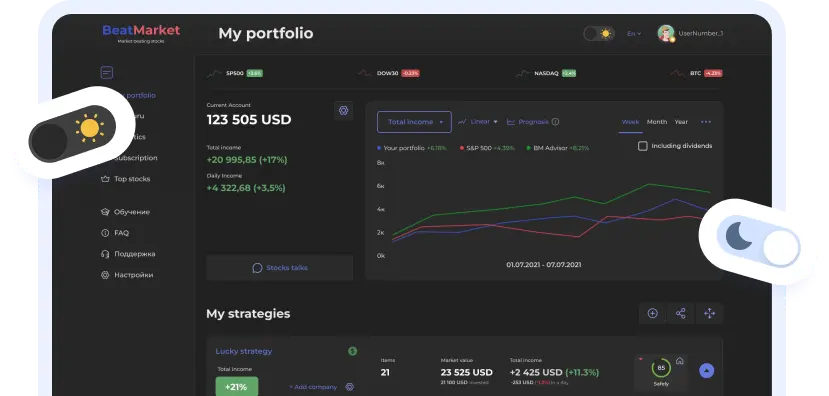



 BeatStart
BeatStart