Valneva SE ADR
VLAValneva SE, a specialty vaccine company, develops, manufactures, and commercializes prophylactic vaccines for infectious diseases with unmet needs. It offers IXIARO, an inactivated Vero cell culture-derived Japanese encephalitis vaccine indicated for active immunization against Japanese encephalitis; DUKORAL, an oral vaccine for the prevention of diarrhea caused by Vibrio cholera and/or heat-labile toxin producing enterotoxigenic Escherichia coli bacterium; IXCHIQ, a single-dose, live-attenuated vaccine for the prevention of disease caused by chikungunya virus; and VLA2001, an inactivated whole-virus COVID-19 vaccine. The company also develops VLA15, a vaccine candidate, which is in Phase III clinical trial against Borrelia, the bacterium that causes Lyme disease; VLA1553, a vaccine candidate, which is in Phase III clinical trial against the chikungunya virus; VLA1554, a vaccine candidate targeting human metapneumovirus; and VLA2112, a vaccine candidate to treat patients with epstein-barr virus. It sells its products in the United States, Canada, Germany, Austria, Nordics, the United Kingdom, France, rest of European countries, and internationally. Valneva SE has collaborations with Pfizer, Inc. to co-develop and commercialize its Lyme disease vaccine; and Instituto Butantan for the development, manufacturing, and marketing of single-shot chikungunya vaccine. The company was founded in 1998 and is based in Saint-Herblain, France. Address: 6 rue Alain Bombard, Saint-Herblain, France, 44800




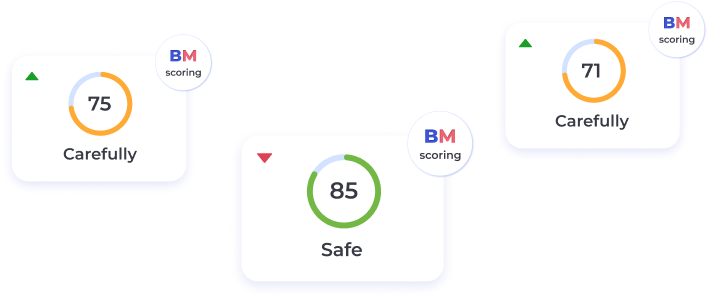


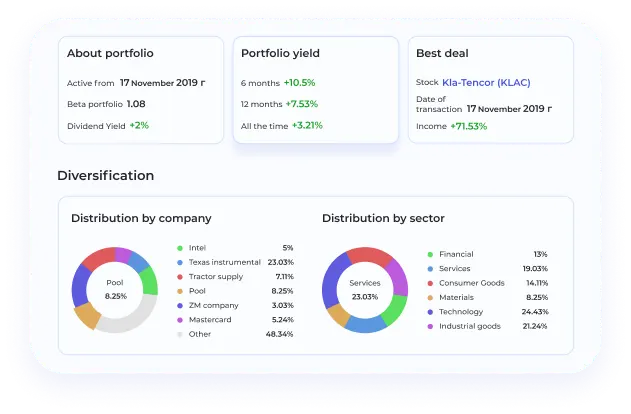


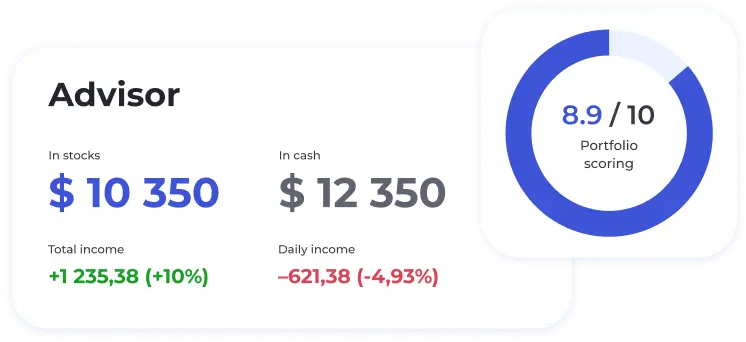
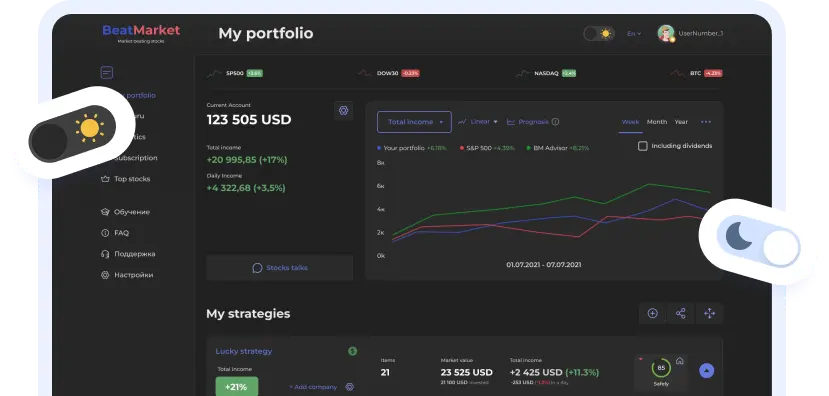



 BeatStart
BeatStart