Introduzca una acción o criptomoneda en el cuadro de búsqueda para obtener un resumen

Cara Therapeutic
69CCara Therapeutics, Inc., a development-stage biopharmaceutical company, focuses on developing and commercializing therapeutics treatment of chronic pruritus in the United States. The company's lead product is KORSUVA (difelikefalin) injection for the treatment of moderate-to-severe pruritus associated with chronic kidney disease (CKD) in adults undergoing hemodialysis. It also develops Oral difelikefalin, which is in Phase II/III clinical trial to treat chronic pruritus with notalgia paresthetica. The company has license agreements with Maruishi Pharmaceutical Co., Ltd to develop, manufacture, and commercialize drug products containing difelikefalin for acute pain and uremic pruritus in Japan; Vifor Fresenius Medical Care Renal Pharma Ltd. development and commercialization of KORSUVA injection for the treatment of moderate-to-severe pruritus in adult patients undergoing hemodialysis; and Chong Kun Dang Pharmaceutical Corporation to develop, manufacture, and commercialize drug products containing difelikefalin in South Korea. Cara Therapeutics, Inc. was incorporated in 2004 and is based in Stamford, Connecticut. Address: 400 Atlantic Street, Stamford, CT, United States, 06901
Analytics
Precio Objetivo de WallStreet
1 USDRelación P/E
–Rentabilidad por dividendo
–Año en curso
Año anterior
Trimestre actual
Último trimestre
Año en curso
Año anterior
Trimestre actual
Último trimestre
Cifras clave 69C
Análisis de dividendos 69C
Aumento del dividendo durante 5 años
–Crecimiento continuo
–Ratio de pago medio en 5 años
–Tendencia del payout 69C
Valoración de la acción 69C
Finanzas 69C

Realice un seguimiento de las métricas clave de cada acción en un solo lugar y sin complicaciones
Hazte premium| Resultados | 2019 | Dinámica |



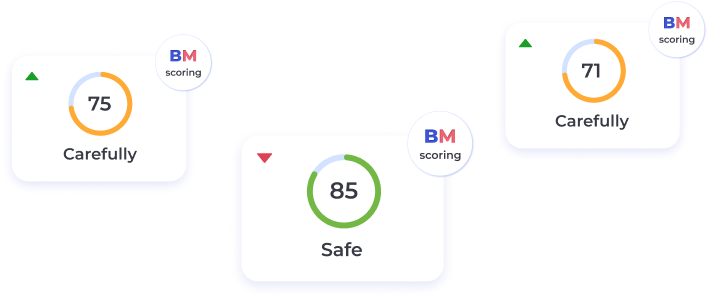

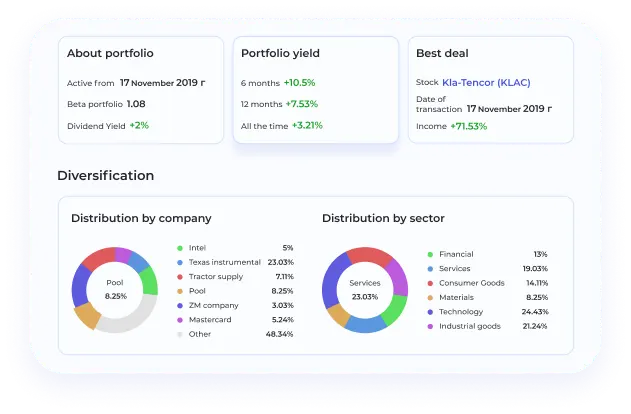


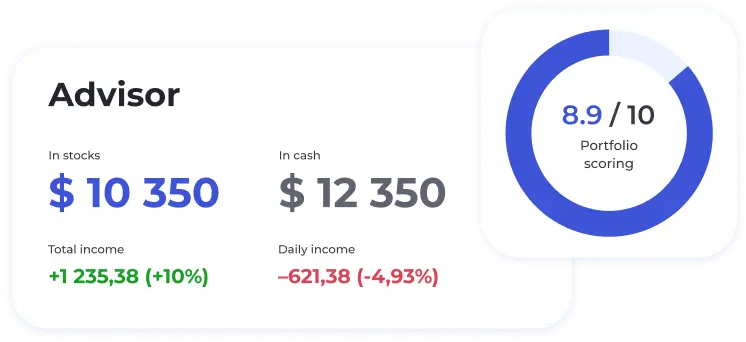
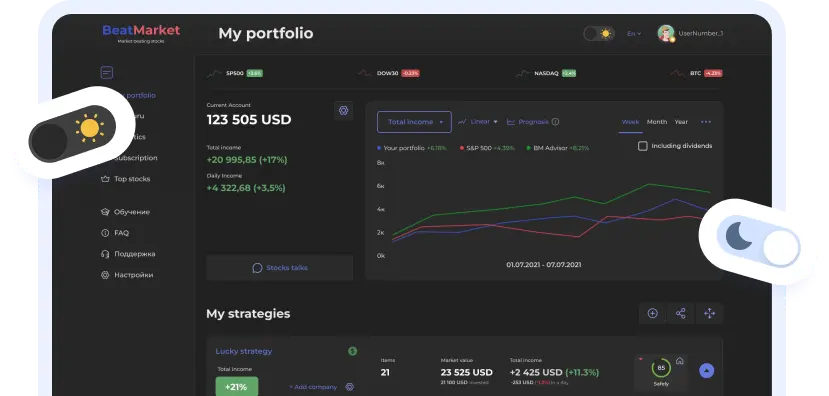



 BeatStart
BeatStart